-
Contact the PP Team
Connect with us
Navigation
Install the app
How to install the app on iOS
Follow along with the video below to see how to install our site as a web app on your home screen.
Note: This feature may not be available in some browsers.
More options
Style variation
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Russia becomes first country to approve a COVID-19 vaccine [Update Post #339]
- Thread starter TrentDevil
- Start date
We, who ?
In my Medical College here in Kerala, India. Main reason is that the Covid+ patients are >50yrs and already have some CV issues.
Syed1
ODI Captain
- Joined
- Jan 22, 2015
- Runs
- 46,041
- Post of the Week
- 3
[MENTION=2099]Cricket[/MENTION]jishial and HCq is not available. Exploiting the situation by the dealers here.As a nation we lack values.
Doc sahab are you treating any covid patients?
If I recall correctly you are at LRH and there a few cases there
Just to share an internal thing from my hospital, My supervisor told us to prescribe it off the record as it not FDA approved and we can face legal problems if something goes against us.
Yeah but as you mentioned it’s a dangerous drug if not properly monitored. We are going to see many HCq poisoning here in India because of this ICMR notification.
We are on duty in corona screening OPD hust once a day in a week . I have not received corona patients yet . My coleagues have received but they came out negative. We are in KTH. KPK is doing very good so far. Tableghi jumaat walo par pabandi lagaani chaahiy . Disgusted with their attitude so far.Doc sahab are you treating any covid patients?
If I recall correctly you are at LRH and there a few cases there
Last edited:
Given dengue itself has no treatment, I would say never. Yes, we can have vaccines but that is about prevention. We may also use certain drugs to control in case of minimal infection spread. But for significant infection, there probably is and will have no cure.
And there is no vaccine for SARS, and virus also can mutate. So its a tough tough battle...
And there is no vaccine for SARS, and virus also can mutate. So its a tough tough battle...
Rana
Hall of Famer
- Joined
- Jan 18, 2006
- Runs
- 90,385
Herd immunity
In my Medical College here in Kerala, India. Main reason is that the Covid+ patients are >50yrs and already have some CV issues.
You're doing the right thing, following " do no harm" . Every treatment we prescribe, its benefit should outweigh the risk. There is no medication developed yet which is 100% free of side effects and every physician has his own comfort level based on studies and experience. After a careful review of patient's medical and medication history, we are using Hydroxychloquin and Azithromycin on almost all the patients being treated for covid 19 infection in hospital and some as out patient too. But this is an off-label treatment and every physician is responsible for his/her prescription and you're right it cannot be recommended for everyone. Many of the commonly prescribed medications carry higher risk of prolonging QTi than these meds.
Last edited:
[MENTION=140234]DRsohail[/MENTION] There is already a case of HCq poisoning in India. A 4yr old is in ICU now.
https://simplicity.in/news-detail.php?id=63256
Also another man died in Arizona because of it.

https://simplicity.in/news-detail.php?id=63256
Also another man died in Arizona because of it.
cricketjoshila
Test Captain
- Joined
- Mar 16, 2011
- Runs
- 48,495
- Post of the Week
- 1
[MENTION=2099]Cricket[/MENTION]jishial and HCq is not available. Exploiting the situation by the dealers here.As a nation we lack values.
People hoarding hcq in india as well.
cricketjoshila
Test Captain
- Joined
- Mar 16, 2011
- Runs
- 48,495
- Post of the Week
- 1
[MENTION=140234]DRsohail[/MENTION] There is already a case of HCq poisoning in India. A 4yr old is in ICU now.
https://simplicity.in/news-detail.php?id=63256
Also another man died in Arizona because of it.
4 yr old? Hiw did he get the drug?
cricketjoshila
Test Captain
- Joined
- Mar 16, 2011
- Runs
- 48,495
- Post of the Week
- 1
You're doing the right thing, following " do no harm" . Every treatment we prescribe, its benefit should outweigh the risk. There is no medication developed yet which is 100% free of side effects and every physician has his own comfort level based on studies and experience. After a careful review of patient's medical and medication history, we are using Hydroxychloquin and Azithromycin on almost all the patients being treated for covid 19 infection in hospital and some as out patient too. But this is an off-label treatment and every physician is responsible for his/her prescription and you're right it cannot be recommended for everyone. Many of the commonly prescribed medications carry higher risk of prolonging QTi than these meds.
How is the progress of patients treated with HCQs and Azithromycin?
JaDed
T20I Captain
- Joined
- May 5, 2014
- Runs
- 41,258
Praying everyday for it, atleast something..
How is the progress of patients treated with HCQs and Azithromycin?
Non-ICU doing better, some of them, ICU pts, not so great.
Rana
Hall of Famer
- Joined
- Jan 18, 2006
- Runs
- 90,385
Don’t think there ever will be one.
It will have to be herd immunity or something that is treated to cure other illnesses.
It will have to be herd immunity or something that is treated to cure other illnesses.
Syed1
ODI Captain
- Joined
- Jan 22, 2015
- Runs
- 46,041
- Post of the Week
- 3
I have this sense of dread that we may never get a full cure and instead may have to 'manage' it like the flu. Countries and governments cannot afford to go into lockdown for months on end. If these 2-3 week lockdowns don't work then some countries might think about lifting them and getting on with life with the constant threat of corona above their heads.
UP
T20I Debutant
- Joined
- Sep 25, 2007
- Runs
- 8,097
I have this sense of dread that we may never get a full cure and instead may have to 'manage' it like the flu. Countries and governments cannot afford to go into lockdown for months on end. If these 2-3 week lockdowns don't work then some countries might think about lifting them and getting on with life with the constant threat of corona above their heads.
Lockdowns really only started a mere week ago and businesses have collapsed and industries are laying of millions of people. Imagine the damage this virus will cause of if businesses stay closed for only 1-2 months.
I think we are in this mess for at least 6-12 months.
This virus could potentially change the way we live for sometime, make people more frugal. And mindful about where they travel and how. The world 6 months from now will be much much different than 6 months ago that's for sure.
I just don't see how governments will allow people to go back to work and resume life as nothing happened while hospital systems all around the world are in a meltdown. This virus must be defeated one way or another, the US is lacking political leadership, we should bar inter state travel aswell until we contain it. The way we're handling it currently we will be in for the long haul because people will continue to travel and spread the virus. Only now did they say everyone who has been in NYC should "self-quarantine" that will not work.
I just don't see how governments will allow people to go back to work and resume life as nothing happened while hospital systems all around the world are in a meltdown. This virus must be defeated one way or another, the US is lacking political leadership, we should bar inter state travel aswell until we contain it. The way we're handling it currently we will be in for the long haul because people will continue to travel and spread the virus. Only now did they say everyone who has been in NYC should "self-quarantine" that will not work.
The problem is long genetic material of virus which is bound to change continuously so Vaccine will be difficult as new strains will be found. The best way would be to find vaccine for the deadliest strains . Some experts think that the cirsu may self destruct itslef by nonsustainable genetic changes or it may become more dangerous.I have this sense of dread that we may never get a full cure and instead may have to 'manage' it like the flu. Countries and governments cannot afford to go into lockdown for months on end. If these 2-3 week lockdowns don't work then some countries might think about lifting them and getting on with life with the constant threat of corona above their heads.
That is good news and may help us .Non-ICU doing better, some of them, ICU pts, not so great.
JaDed
T20I Captain
- Joined
- May 5, 2014
- Runs
- 41,258
Checking news everyday anxiously and anything looks unlikely...
All hope on virus to not mutate and destroy itself!
All hope on virus to not mutate and destroy itself!
Varun
Senior Test Player
- Joined
- Dec 25, 2012
- Runs
- 26,320
- Post of the Week
- 1
Checking news everyday anxiously and anything looks unlikely...
All hope on virus to not mutate and destroy itself!
Hopefully the summer will help reduce the impact.
*Information quarantined village at Mardan, KPK:*
Quick update of extended Corona Virus test results in UC Manga, Mardan where Pakistan's first death was recorded, & a lockdown was enacted.
Sampling has shown that out of 46 people tested in the UC, 39 tests are positive.
4 samples were rejected. All tested were asymptomatic.
Hence, please exercise caution in your daily day to day routine public dealings to maximum possible in Isbd as well as it can affect you, your families and even communities where you reside.
Fyi.
Quick update of extended Corona Virus test results in UC Manga, Mardan where Pakistan's first death was recorded, & a lockdown was enacted.
Sampling has shown that out of 46 people tested in the UC, 39 tests are positive.
4 samples were rejected. All tested were asymptomatic.
Hence, please exercise caution in your daily day to day routine public dealings to maximum possible in Isbd as well as it can affect you, your families and even communities where you reside.
Fyi.
- Joined
- Oct 2, 2004
- Runs
- 218,134
Millions of new coronavirus testing kits could be ready to order on Amazon in days, rather than weeks or months, according to Public Health England.
Prime Minister Boris Johnson has previously spoken of a simple test for coronavirus as a potential "total game changer" in the battle against COVID-19.
Professor Sharon Peacock, director of the National Infection Service at Public Health England, revealed on Wednesday that millions of such a test have been ordered and could be used "in the near future".
They would show whether an individual had antibodies for COVID-19 which, if they did, would mean they could return to work if they were not showing symptoms.
Evidence suggests people cannot catch coronavirus twice in quick succession, if they have already been infected and recovered, the government has said.
The tests, which look similar to pregnancy tests, could be ordered on Amazon or taken at high street branches such as Boots, Prof Peacock said.
The World Health Organisation has urged countries to step up testing in the global fight against coronavirus.
Appearing before the House of Commons science and technology committee via video link, Prof Peacock said the new tests were "rapidly" being evaluated at a laboratory in Oxford with 3.5 million having already been ordered.
"Further millions are being ordered today," Prof Peacock told MPs.
"But we do need to ensure we understand how they operate, because these are brand new tests."
Prof Peacock revealed the evaluation of the tests would likely be done by the end of this week.
She added: "In the near future people will be able to order a test so they can test themselves or go to Boots or somewhere similar to have their finger-***** test done."
Asked if the availability of the test to the public would be a number of days rather than weeks or months, she replied: "Absolutely."
Jeremy Hunt asks the prime minister about coronavirus testing
'Should we introduce weekly tests for NHS staff?'
Prof Peacock told the committee there are "two different models", with one possible model being a test that is ordered via Amazon and performed at home before being sent back to see whether someone tests positive or negative for coronavirus.
Another "might require you to go somewhere like Boots because it requires a blood *****", she added.
Explaining further how the tests would work, she continued: "It looks like a pregnancy test. Except that you're putting a finger with a spot of blood on there.
"You ***** your finger like a diabetic would, then get a drop of blood and put it on a filter paper and then run some liquid to make that blood run into the test zone."
Public Health England will be asking some people to have a second blood test, checked with a "gold standard" test, to make sure the kits are working properly, Prof Peacock said.
The tests "can see if you have antibodies, in which case you know you had the infection", she added.
"This is not just for key workers, its for the general population. Over time its expected the proportion of the population will be positive.
"And that will allow them to get back to work."
Prof Peacock said she couldn't comment on whether the tests would be charged for but predicted there "would be an absolutely minimal charge if there was a charge".
https://news.sky.com/story/coronavi...-could-be-available-in-days-mps-told-11963509
Prime Minister Boris Johnson has previously spoken of a simple test for coronavirus as a potential "total game changer" in the battle against COVID-19.
Professor Sharon Peacock, director of the National Infection Service at Public Health England, revealed on Wednesday that millions of such a test have been ordered and could be used "in the near future".
They would show whether an individual had antibodies for COVID-19 which, if they did, would mean they could return to work if they were not showing symptoms.
Evidence suggests people cannot catch coronavirus twice in quick succession, if they have already been infected and recovered, the government has said.
The tests, which look similar to pregnancy tests, could be ordered on Amazon or taken at high street branches such as Boots, Prof Peacock said.
The World Health Organisation has urged countries to step up testing in the global fight against coronavirus.
Appearing before the House of Commons science and technology committee via video link, Prof Peacock said the new tests were "rapidly" being evaluated at a laboratory in Oxford with 3.5 million having already been ordered.
"Further millions are being ordered today," Prof Peacock told MPs.
"But we do need to ensure we understand how they operate, because these are brand new tests."
Prof Peacock revealed the evaluation of the tests would likely be done by the end of this week.
She added: "In the near future people will be able to order a test so they can test themselves or go to Boots or somewhere similar to have their finger-***** test done."
Asked if the availability of the test to the public would be a number of days rather than weeks or months, she replied: "Absolutely."
Jeremy Hunt asks the prime minister about coronavirus testing
'Should we introduce weekly tests for NHS staff?'
Prof Peacock told the committee there are "two different models", with one possible model being a test that is ordered via Amazon and performed at home before being sent back to see whether someone tests positive or negative for coronavirus.
Another "might require you to go somewhere like Boots because it requires a blood *****", she added.
Explaining further how the tests would work, she continued: "It looks like a pregnancy test. Except that you're putting a finger with a spot of blood on there.
"You ***** your finger like a diabetic would, then get a drop of blood and put it on a filter paper and then run some liquid to make that blood run into the test zone."
Public Health England will be asking some people to have a second blood test, checked with a "gold standard" test, to make sure the kits are working properly, Prof Peacock said.
The tests "can see if you have antibodies, in which case you know you had the infection", she added.
"This is not just for key workers, its for the general population. Over time its expected the proportion of the population will be positive.
"And that will allow them to get back to work."
Prof Peacock said she couldn't comment on whether the tests would be charged for but predicted there "would be an absolutely minimal charge if there was a charge".
https://news.sky.com/story/coronavi...-could-be-available-in-days-mps-told-11963509
It appears the virus mutates slowly, so a vaccine would provide lasting immunity as opposed to the seasonal flu vaccine.
https://www.washingtonpost.com/heal...6522d6-6dfd-11ea-b148-e4ce3fbd85b5_story.html
https://www.washingtonpost.com/heal...6522d6-6dfd-11ea-b148-e4ce3fbd85b5_story.html
Last edited:
It appears the virus mutates slowly, so a vaccine would provide lasting immunity as opposed to the seasonal flu vaccine.
https://www.washingtonpost.com/heal...6522d6-6dfd-11ea-b148-e4ce3fbd85b5_story.html
Thanks, its different from the information he had last week, good news if proved to be true.
cricketjoshila
Test Captain
- Joined
- Mar 16, 2011
- Runs
- 48,495
- Post of the Week
- 1
Thanks, its different from the information he had last week, good news if proved to be true.
Tbh we really dont know much. We have new conflicting info each week. Having a bumbling mumbling bufoon as WHO DG isn't helping.
As frontline responders we are like soldiers walking into enemy territory blindfolded with no intel and not knowing if our weapons are effective.
Abdul
ODI Debutant
- Joined
- Jul 1, 2015
- Runs
- 9,212
Hopefully the summer will help reduce the impact.
It won't. It's summer in Australia right now and there are lots of cases there.
cricketjoshila
Test Captain
- Joined
- Mar 16, 2011
- Runs
- 48,495
- Post of the Week
- 1
It won't. It's summer in Australia right now and there are lots of cases there.
Actually Australia is entering winter.
last_knight
T20I Debutant
- Joined
- Jan 23, 2012
- Runs
- 6,461
cricketjoshila
Test Captain
- Joined
- Mar 16, 2011
- Runs
- 48,495
- Post of the Week
- 1
Covid-19 is still in early days with more and more data coming every day.
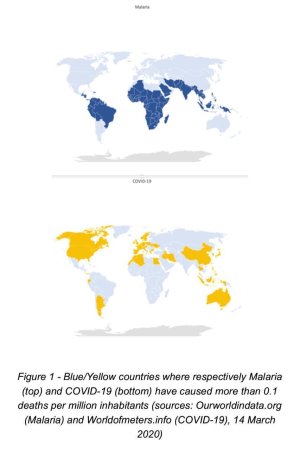
It's too early to say something but this comparison may not be just another coincidence :
View attachment 100160
May be not a coincidence.
shebimalik
Tape Ball Star
- Joined
- Dec 24, 2015
- Runs
- 903
<blockquote class="twitter-tweet"><p lang="en" dir="ltr">Chinese scientists have developed a new weapon to combat the <a href="https://twitter.com/hashtag/coronavirus?src=hash&ref_src=twsrc%5Etfw">#coronavirus</a>. They say they have found a nanomaterial that can absorb and deactivate the virus with 96.5-99.9% efficiency. <a href="https://t.co/ESFUOoTuIX">pic.twitter.com/ESFUOoTuIX</a></p>— Global Times (@globaltimesnews) <a href="https://twitter.com/globaltimesnews/status/1244244793393217536?ref_src=twsrc%5Etfw">March 29, 2020</a></blockquote> <script async src="https://platform.twitter.com/widgets.js" charset="utf-8"></script>
Covid-19 is still in early days with more and more data coming every day.
It's too early to say something but this comparison may not be just another coincidence :
View attachment 100160
What about Iran ? Yellow and Blue ?
Syed1
ODI Captain
- Joined
- Jan 22, 2015
- Runs
- 46,041
- Post of the Week
- 3
<blockquote class="twitter-tweet"><p lang="en" dir="ltr">Chinese scientists have developed a new weapon to combat the <a href="https://twitter.com/hashtag/coronavirus?src=hash&ref_src=twsrc%5Etfw">#coronavirus</a>. They say they have found a nanomaterial that can absorb and deactivate the virus with 96.5-99.9% efficiency. <a href="https://t.co/ESFUOoTuIX">pic.twitter.com/ESFUOoTuIX</a></p>— Global Times (@globaltimesnews) <a href="https://twitter.com/globaltimesnews/status/1244244793393217536?ref_src=twsrc%5Etfw">March 29, 2020</a></blockquote> <script async src="https://platform.twitter.com/widgets.js" charset="utf-8"></script>
Chimgaadar ke khoon se hi bunaya hona hai in Chini-yoon ne
CricketCartoons
Senior T20I Player
- Joined
- Mar 5, 2014
- Runs
- 17,584
September will be the earliest when the vaccine can be available to the public, at least in US.
- Joined
- Oct 2, 2004
- Runs
- 218,134
LAHORE: The Punjab government has allowed treatment of critically ill patients of Covid-19 with chloroquine and hydroxychloroquine besides expanding the capacity of beds across the province.
The government has procured 50,000 tablets of the two said medicines.
Mayo Hospital had earlier started using the drug on critical patients of Covid-19. China had also treated its critical patients suffering from the virus and the US has also allowed its use.
The decision was taken by the government after the Food and Drug Authority (FDA) of the US granted approval to using hydroxychloroquine, which is originally used to treat malaria.
“Our physicians have started medication of the seriously ill patients of coronavirus at the Mayo Hospital and initial reports suggest that the drug is yielding positive results,” unveiled Mayo Hospital Lahore Chief Executive Prof Dr Asad Aslam Khan.
Two three-star hotels to be rented to isolate suspected cases
Talking to Dawn, he said the Mayo physicians were suggesting treatment of Covid-19 patients through chloroquine and hydroxychloroquine, which had been provided in sufficient quantity by the Punjab government.
Meanwhile, the government planned to rent two three-star hotels in the city to use their rooms to isolate suspected patients of coronavirus.
It has also given permission to start outdoor services for suspected patients of coronavirus at the Mayo Hospital and the field hospital at Expo Centre. The decision was taken following a surge in the number of Covid-19 cases across the province.
The 1,000-bed Expo Centre Hospital has also been completed for patient management and will start functioning soon after the recruitment of 1,900 doctors, nurses and paramedics.
On Monday, Dr Khan said, Health Minister Dr Yasmin Rashid reviewed final arrangements at the Expo Centre and addressed the staff, including doctors, to motivate them for this daunting task. Dr Khan has also been appointed as administrator of the Expo Centre Hospital.
“We have divided the 1,000-bed Expo Centre Hospital into three portions for the patients,” he explained.
Two portions will be used to isolate mild and severe patients of the disease separately. The third potion will be used for seriously ill patients, he said, adding that a 50-bed high dependency unit had also been made fully functional with ventilators.
“We have deputed highly trained medics and other staff for triage site at the Expo Centre, while Rescue 1122 has been hired to shift patients to this facility from all over the province,” he added.
He further said that Fatima Jinnah Medical University Vice Chancellor Prof Dr Aamir Zeman Khan was conducting interviews to recruit 1,900 doctors, nurses and paramedics for the Expo Centre Hospital.
“For the first time, we are going to start OPD service for suspected coronavirus patients only [at Mayo and Expo Centre hospitals],” said Dr Khan.
He also said the Mayo Hospital OPD has become the largest set-up for coronavirus in Punjab with 500 more beds added to the isolation and quarantine wards here. The patients testing positive at the OPD here would be shifted to the Expo Centre Hospital, he added.
https://www.dawn.com/news/1545099/p...ria-drug-for-critically-ill-covid-19-patients
The government has procured 50,000 tablets of the two said medicines.
Mayo Hospital had earlier started using the drug on critical patients of Covid-19. China had also treated its critical patients suffering from the virus and the US has also allowed its use.
The decision was taken by the government after the Food and Drug Authority (FDA) of the US granted approval to using hydroxychloroquine, which is originally used to treat malaria.
“Our physicians have started medication of the seriously ill patients of coronavirus at the Mayo Hospital and initial reports suggest that the drug is yielding positive results,” unveiled Mayo Hospital Lahore Chief Executive Prof Dr Asad Aslam Khan.
Two three-star hotels to be rented to isolate suspected cases
Talking to Dawn, he said the Mayo physicians were suggesting treatment of Covid-19 patients through chloroquine and hydroxychloroquine, which had been provided in sufficient quantity by the Punjab government.
Meanwhile, the government planned to rent two three-star hotels in the city to use their rooms to isolate suspected patients of coronavirus.
It has also given permission to start outdoor services for suspected patients of coronavirus at the Mayo Hospital and the field hospital at Expo Centre. The decision was taken following a surge in the number of Covid-19 cases across the province.
The 1,000-bed Expo Centre Hospital has also been completed for patient management and will start functioning soon after the recruitment of 1,900 doctors, nurses and paramedics.
On Monday, Dr Khan said, Health Minister Dr Yasmin Rashid reviewed final arrangements at the Expo Centre and addressed the staff, including doctors, to motivate them for this daunting task. Dr Khan has also been appointed as administrator of the Expo Centre Hospital.
“We have divided the 1,000-bed Expo Centre Hospital into three portions for the patients,” he explained.
Two portions will be used to isolate mild and severe patients of the disease separately. The third potion will be used for seriously ill patients, he said, adding that a 50-bed high dependency unit had also been made fully functional with ventilators.
“We have deputed highly trained medics and other staff for triage site at the Expo Centre, while Rescue 1122 has been hired to shift patients to this facility from all over the province,” he added.
He further said that Fatima Jinnah Medical University Vice Chancellor Prof Dr Aamir Zeman Khan was conducting interviews to recruit 1,900 doctors, nurses and paramedics for the Expo Centre Hospital.
“For the first time, we are going to start OPD service for suspected coronavirus patients only [at Mayo and Expo Centre hospitals],” said Dr Khan.
He also said the Mayo Hospital OPD has become the largest set-up for coronavirus in Punjab with 500 more beds added to the isolation and quarantine wards here. The patients testing positive at the OPD here would be shifted to the Expo Centre Hospital, he added.
https://www.dawn.com/news/1545099/p...ria-drug-for-critically-ill-covid-19-patients
Kroll
ODI Debutant
- Joined
- Jun 4, 2017
- Runs
- 10,274
- Post of the Week
- 1
Coronavirus: Australian scientists begin tests of potential vaccines
Scientists in Australia have begun testing two potential coronavirus vaccines in "milestone" lab trials.
The vaccines, made by Oxford University and US company Inovio Pharmaceutical, have been cleared for animal testing by the World Health Organization.
Australia's national science agency will assess if the vaccines work, and if they would be safe for humans.
The first human trial took place in the US last month, but skipped a stage of animal testing.
There are several other vaccine developments occurring around the world at the moment at extraordinary speed.
But Australia's Commonwealth Scientific and Industrial Research Organisation (CSIRO) says its tests will be the first comprehensive pre-clinical trials of the vaccines to use an animal model.
Researchers said the speed and level of global co-operation that led to this stage had been unprecedented.
"Normally it takes about one-to-two years to get to this point and we've in fact shortened that to a period of a couple of months," Dr Rob Grenfell from the CSIRO told reporters on Thursday.
How will it work?
In the past few days, the CSIRO team has inserted vaccine samples into ferrets - small, furry mammals which have been proven to contract the coronavirus in the same way humans do.
Sars-CoV-2 is the virus which causes the disease Covid-19. There are at least 20 vaccines in development around the world.
The CSIRO is testing two options selected by a global consortium overseeing most of this research, the Coalition for Epidemic Preparedness Innovations.
One option, developed by the University of Oxford, is a vector vaccine. It uses a "defective" virus to introduce the proteins of the coronavirus to the immune system and induce a response.
"But they're not able to replicate... so there's no possibility of becoming ill through this particular vaccine," said Prof Trevor Drew, director of the Australian Animal Health Laboratory in Victoria, where the testing is being carried out.
He described the other vaccine - from Inovio - as "rather different but nonetheless exciting".
It is designed to encode certain proteins of the coronavirus to the immune system, prompting the body's cells to generate those proteins before the immune system reacts to them.
"It's very important to take a multi-pronged approach to this," said Prof Drew. "It gives us the best chance of success."
How soon could we get results?
The first results from the animal tests could be revealed as early as June, the scientists said.
If successful, the vaccines could then be moved into clinical trials that could be conducted at labs elsewhere.
At that point, the race to get the medicine into the general market could be accelerated, but experts warn it would still involve a minimum 18-month timeline to meet regulatory tests and standards.
The Oxford University vaccine, for example, is also being tested by another lab on a separate animal to comply with US regulations.
"All of the vaccine candidates actually have a multitude of other companies and organisations involved because no single organisation can actually produce a vaccine," said Dr Grenfell.
But he said he was "optimistic" about the work, given the level of global collaboration.
"This is a true collaboration across academic, public, and also private sectors to in fact actually achieve success at this point," he said.
https://www.bbc.com/news/world-australia-52130402
Scientists in Australia have begun testing two potential coronavirus vaccines in "milestone" lab trials.
The vaccines, made by Oxford University and US company Inovio Pharmaceutical, have been cleared for animal testing by the World Health Organization.
Australia's national science agency will assess if the vaccines work, and if they would be safe for humans.
The first human trial took place in the US last month, but skipped a stage of animal testing.
There are several other vaccine developments occurring around the world at the moment at extraordinary speed.
But Australia's Commonwealth Scientific and Industrial Research Organisation (CSIRO) says its tests will be the first comprehensive pre-clinical trials of the vaccines to use an animal model.
Researchers said the speed and level of global co-operation that led to this stage had been unprecedented.
"Normally it takes about one-to-two years to get to this point and we've in fact shortened that to a period of a couple of months," Dr Rob Grenfell from the CSIRO told reporters on Thursday.
How will it work?
In the past few days, the CSIRO team has inserted vaccine samples into ferrets - small, furry mammals which have been proven to contract the coronavirus in the same way humans do.
Sars-CoV-2 is the virus which causes the disease Covid-19. There are at least 20 vaccines in development around the world.
The CSIRO is testing two options selected by a global consortium overseeing most of this research, the Coalition for Epidemic Preparedness Innovations.
One option, developed by the University of Oxford, is a vector vaccine. It uses a "defective" virus to introduce the proteins of the coronavirus to the immune system and induce a response.
"But they're not able to replicate... so there's no possibility of becoming ill through this particular vaccine," said Prof Trevor Drew, director of the Australian Animal Health Laboratory in Victoria, where the testing is being carried out.
He described the other vaccine - from Inovio - as "rather different but nonetheless exciting".
It is designed to encode certain proteins of the coronavirus to the immune system, prompting the body's cells to generate those proteins before the immune system reacts to them.
"It's very important to take a multi-pronged approach to this," said Prof Drew. "It gives us the best chance of success."
How soon could we get results?
The first results from the animal tests could be revealed as early as June, the scientists said.
If successful, the vaccines could then be moved into clinical trials that could be conducted at labs elsewhere.
At that point, the race to get the medicine into the general market could be accelerated, but experts warn it would still involve a minimum 18-month timeline to meet regulatory tests and standards.
The Oxford University vaccine, for example, is also being tested by another lab on a separate animal to comply with US regulations.
"All of the vaccine candidates actually have a multitude of other companies and organisations involved because no single organisation can actually produce a vaccine," said Dr Grenfell.
But he said he was "optimistic" about the work, given the level of global collaboration.
"This is a true collaboration across academic, public, and also private sectors to in fact actually achieve success at this point," he said.
https://www.bbc.com/news/world-australia-52130402
BoredFromLifeXi
Debutant
- Joined
- Jan 28, 2020
- Runs
- 77
A Question regarding testing kit for COVID 19
Hello Folks, I read somewhere on internet that RT-PCR is not highly accurate test for coronvirus detection. It has efficiency around only 90%
I have a question then, What test method are used by countries around the world if this has 10% chance of getting wrong result. I create a new thread because it's important to know what is following around a world. Can any doctor here will clear it more. Thank you
Hello Folks, I read somewhere on internet that RT-PCR is not highly accurate test for coronvirus detection. It has efficiency around only 90%
I have a question then, What test method are used by countries around the world if this has 10% chance of getting wrong result. I create a new thread because it's important to know what is following around a world. Can any doctor here will clear it more. Thank you
BoredFromLifeXi
Debutant
- Joined
- Jan 28, 2020
- Runs
- 77
And do RT-PCR test was made only for research purpose, not diagnostic purpose?
- Joined
- Oct 2, 2004
- Runs
- 218,134
PESHAWAR: The Khyber Pakhtunkhwa health department has allowed use of plasma extracted from the blood of patients having fully recovered from Covid-19 for the treatment of the fresh cases.
A notification issued from the office of secretary health on Sunday said the Provincial Passive Immunisation Committee proposed by the Hayatabad Medical Complex and comprising health experts and professionals had been given a go-ahead to submit a detailed proposal on the subject for onward submission to the National Bioethics Committee for approval.
On March 28, the Hayatabad Medical Complex’s medical director Prof Shehzad Akbar Khan had requested the health secretary to constitute a committee in accordance with a proposal made by Prof Tahir Shamsi, chairman of the National Passive Immunisation Committee, which had also been approved the US Food and Drug Administration, to treat the fresh patients with plasma extracted from the blood of patients having fully recovered from the disease.
On Friday, the National Bioethics Committee had approved passive immunisation trial by the HMC but waited for the provincial government’s formal approval of the committee to start treatment of coronavirus patients by plasma.
Meanwhile, health minister Taimur Khan Jhagra in a tweet said the committee had been notified to look at and expedite work on passive immunisation.
https://www.dawn.com/news/1546764/use-of-plasma-allowed-for-treating-covid-19-patients-in-kp
A notification issued from the office of secretary health on Sunday said the Provincial Passive Immunisation Committee proposed by the Hayatabad Medical Complex and comprising health experts and professionals had been given a go-ahead to submit a detailed proposal on the subject for onward submission to the National Bioethics Committee for approval.
On March 28, the Hayatabad Medical Complex’s medical director Prof Shehzad Akbar Khan had requested the health secretary to constitute a committee in accordance with a proposal made by Prof Tahir Shamsi, chairman of the National Passive Immunisation Committee, which had also been approved the US Food and Drug Administration, to treat the fresh patients with plasma extracted from the blood of patients having fully recovered from the disease.
On Friday, the National Bioethics Committee had approved passive immunisation trial by the HMC but waited for the provincial government’s formal approval of the committee to start treatment of coronavirus patients by plasma.
Meanwhile, health minister Taimur Khan Jhagra in a tweet said the committee had been notified to look at and expedite work on passive immunisation.
https://www.dawn.com/news/1546764/use-of-plasma-allowed-for-treating-covid-19-patients-in-kp
Uzi
First Class Captain
- Joined
- Dec 7, 2004
- Runs
- 4,484
Guys, does Ivermectin has any effect on Covid patients???
This is being researched currently, but it is in the very early stages. Ivermectin has not been tested on people yet.
In lab testing it has been shown to shutdown viral replication within 24 to 48 hours. However it is still very early days in testing .
I guess the one good thing is that re-purposing Ivermectin as a potential treatment for C-19 would be ideal as it can move quickly to clinical trial testing as we know it is safe for humans.
Abdullah719
T20I Captain
- Joined
- Apr 16, 2013
- Runs
- 44,825
<blockquote class="twitter-tweet" data-lang="en"><p lang="en" dir="ltr">French hospital halts hydroxychloroquine treatment study due to cardiac side effects <a href="https://t.co/jCkjyH2CDO">https://t.co/jCkjyH2CDO</a></p>— Josh "Wash Your Elbows" Gerstein (@joshgerstein) <a href="https://twitter.com/joshgerstein/status/1247682413687222272?ref_src=twsrc%5Etfw">April 8, 2020</a></blockquote>
<script async src="https://platform.twitter.com/widgets.js" charset="utf-8"></script>
<script async src="https://platform.twitter.com/widgets.js" charset="utf-8"></script>
last_knight
T20I Debutant
- Joined
- Jan 23, 2012
- Runs
- 6,461
Guys, does Ivermectin has any effect on Covid patients???
[MENTION=76058]cricketjoshila[/MENTION]
[MENTION=141839]moghul[/MENTION]
[MENTION=128087]last_knight[/MENTION]
[MENTION=140234]DRsohail[/MENTION]
I will be in a better position to comment after first hand knowledge/experience ( which isn't very likely).
I don't know anyone who is using it for Covid-19. All I know about its use in Covid-19 is from social media news.
Guys, does Ivermectin has any effect on Covid patients???
[MENTION=76058]cricketjoshila[/MENTION]
[MENTION=141839]moghul[/MENTION]
[MENTION=128087]last_knight[/MENTION]
[MENTION=140234]DRsohail[/MENTION]
It has shown some promising results invitro ( in labs only ) but as far as I know, it has not been tried on Covid 19 patients,
<blockquote class="twitter-tweet" data-lang="en"><p lang="en" dir="ltr">French hospital halts hydroxychloroquine treatment study due to cardiac side effects <a href="https://t.co/jCkjyH2CDO">https://t.co/jCkjyH2CDO</a></p>— Josh "Wash Your Elbows" Gerstein (@joshgerstein) <a href="https://twitter.com/joshgerstein/status/1247682413687222272?ref_src=twsrc%5Etfw">April 8, 2020</a></blockquote>
<script async src="https://platform.twitter.com/widgets.js" charset="utf-8"></script>
Hydroxychloquine has shown promise but has not been PROVEN to be curative for covid 19 infection. We have been using on almost every hospitalized covid 19 patient. When used without any other medication which can cause heart problem ( arrhythmia due to prolonged QTc) we have not seen any patient any problem with this meds and we have used on hundreds of patients. When used with other meds including Azithromycin or psych medications risk is more and have to be careful.
But this drug is not a candy, can cause side effects and no one should take it without prescription from his/her doctor. I emphasized this also in my interview with a TV Channel in Pakistan 2 weeks ago.
Last edited:
- Joined
- Oct 2, 2004
- Runs
- 218,134
From my younger days in Pakistan, I can tell you that this medicine was vile! made you so nauseous!
cricketjoshila
Test Captain
- Joined
- Mar 16, 2011
- Runs
- 48,495
- Post of the Week
- 1
Guys, does Ivermectin has any effect on Covid patients???
[MENTION=76058]cricketjoshila[/MENTION]
[MENTION=141839]moghul[/MENTION]
[MENTION=128087]last_knight[/MENTION]
[MENTION=140234]DRsohail[/MENTION]
A research in UK says Ivermectin stopped corona virus replication within 48hrs in lab conditions.
Savak
World Star
- Joined
- Feb 16, 2006
- Runs
- 50,940
- Post of the Week
- 3
What if this virus or disease is untreatable?
last_knight
T20I Debutant
- Joined
- Jan 23, 2012
- Runs
- 6,461
It has shown some promising results invitro ( in labs only ) but as far as I know, it has not been tried on Covid 19 patients,
[MENTION=141839]moghul[/MENTION] bro, I am still curious to know if anyone has conducted a trial/Lab Study of Plasmapharesis or Anti-Venom ?
Have you heard about any ?
Romali_rotti
Senior ODI Player
- Joined
- Feb 10, 2005
- Runs
- 22,045
What if this virus or disease is untreatable?
Body will eventually build immunity to the virus, however it is a matter of how long that is going to take.
last_knight
T20I Debutant
- Joined
- Jan 23, 2012
- Runs
- 6,461
What if this virus or disease is untreatable?
Most of times, cure exists already. And if we fail to discover it, nature reveals it in most unexpected ways.
As a species, Humans are much more resilient than any given virus.
Syed1
ODI Captain
- Joined
- Jan 22, 2015
- Runs
- 46,041
- Post of the Week
- 3
Most of times, cure exists already. And if we fail to discover it, nature reveals it in most unexpected ways.
As a species, Humans are much more resilient than any given virus.
There is no cure for HIV or polio
- Joined
- Oct 2, 2004
- Runs
- 218,134
PESHAWAR: The US National Institute of Health has approved a proposal of medical teaching institution Ayub Medical College and Ayub Teaching Hospital Abbottabad to conduct trial on Covid-19 patients to find out treatment for the pandemic, which has no cure so far.
The trial entitled “effectiveness of hydroxychloroquine in Covid-19 patients” was approved by the US National Library of Medicine of National Institute of Health (NIH), last month to try different set of medicines for the treatment of Covid-19 patients.
Prof Umar Farooq, dean and chief executive officer of Ayub Medical College (AMC) and Ayub Teaching Hospital (ATH) Abbottabad, who will lead the trial, said that they had begun work and were hopeful of success.
“There is no timeline about completion of trial as it depends on the number of patients we receive. Approval of our proposal is indication of the ability of our team and its success would benefit the entire world,” he said.
Hydroxychloroquine trial has been approved by US National Institute of Health
Prof Umar said that the trial was meant to find the effectiveness of hydroxychloroquine alone and adjuvant with azithromycin in mild to severe Covid-19 pneumonia patients admitted to their hospital.
The disease has been killing people at alarming speed and 23 trials are currently underway and some encouraging results from Wuhan, China have been recorded.
The drugs being tried are approved for malaria treatment by Food and Drug Administration (FDA), the US, yet their effectiveness in treatment of Covid-19 pneumonia is a question mark owing to which trials are underway in many countries, including the USA, India and China where the pandemic first occurred.
Prof Umar, who is PhD in public health, said that the disease was new and therefore researches were in progress to closely monitor the Covid-19 patients and their treatment and find out effectiveness of the drug.
Soon after the onset of the outbreak of pandemic in China, there has been worldwide demand for treatment of the Covid-19, which has so far killed more than 50,000 people. “It has no treatment as of now but we are sure that our trial will succeed and Pakistan will become a country to be remembered in the history of medicine,” he said.
Prof Umar said that their proposal was approved last month and they began the trial a week ago. “So far, three of our patients have recovered, details about them couldn’t be made public at this point because of the confidentiality of the patients,” he added.
He said that they were distributing patients in groups and they were given three different regimes of drugs to find accurate treatment of Covid-19. Three groups of 25 patients each are getting different medication and the result will be shared with the NIH, the US on completion of the trial on total 75 patients.
According to him, one group of patients will be given a combination of two drugs including Hydroxy-chloroquine and Azithromycin while the second group will receive only Hydroxychloroquine and the third will undergo traditional method of treatment.
“We are taking consent of each and every patient included in the trial and informing them about the research,” said Prof Umar. He said that all information of patients along with their signatures would be sent to the NIH on completion of the trial.
He said that patients for the trial were being selected on the basis of intensity of their diseases and ages. “We include only Covid-19 patients below 50 years. Those with heart, kidney and other serious ailments aren’t allowed to be included in the trial,” he added.
Prof Umar said that a total of 15 doctors working in different specialties and faculties at AMC and ATH were taking part in the process to win honour for the country.
https://www.dawn.com/news/1547911/abbottabad-hospital-to-conduct-drug-trials-on-covid-19-patients
The trial entitled “effectiveness of hydroxychloroquine in Covid-19 patients” was approved by the US National Library of Medicine of National Institute of Health (NIH), last month to try different set of medicines for the treatment of Covid-19 patients.
Prof Umar Farooq, dean and chief executive officer of Ayub Medical College (AMC) and Ayub Teaching Hospital (ATH) Abbottabad, who will lead the trial, said that they had begun work and were hopeful of success.
“There is no timeline about completion of trial as it depends on the number of patients we receive. Approval of our proposal is indication of the ability of our team and its success would benefit the entire world,” he said.
Hydroxychloroquine trial has been approved by US National Institute of Health
Prof Umar said that the trial was meant to find the effectiveness of hydroxychloroquine alone and adjuvant with azithromycin in mild to severe Covid-19 pneumonia patients admitted to their hospital.
The disease has been killing people at alarming speed and 23 trials are currently underway and some encouraging results from Wuhan, China have been recorded.
The drugs being tried are approved for malaria treatment by Food and Drug Administration (FDA), the US, yet their effectiveness in treatment of Covid-19 pneumonia is a question mark owing to which trials are underway in many countries, including the USA, India and China where the pandemic first occurred.
Prof Umar, who is PhD in public health, said that the disease was new and therefore researches were in progress to closely monitor the Covid-19 patients and their treatment and find out effectiveness of the drug.
Soon after the onset of the outbreak of pandemic in China, there has been worldwide demand for treatment of the Covid-19, which has so far killed more than 50,000 people. “It has no treatment as of now but we are sure that our trial will succeed and Pakistan will become a country to be remembered in the history of medicine,” he said.
Prof Umar said that their proposal was approved last month and they began the trial a week ago. “So far, three of our patients have recovered, details about them couldn’t be made public at this point because of the confidentiality of the patients,” he added.
He said that they were distributing patients in groups and they were given three different regimes of drugs to find accurate treatment of Covid-19. Three groups of 25 patients each are getting different medication and the result will be shared with the NIH, the US on completion of the trial on total 75 patients.
According to him, one group of patients will be given a combination of two drugs including Hydroxy-chloroquine and Azithromycin while the second group will receive only Hydroxychloroquine and the third will undergo traditional method of treatment.
“We are taking consent of each and every patient included in the trial and informing them about the research,” said Prof Umar. He said that all information of patients along with their signatures would be sent to the NIH on completion of the trial.
He said that patients for the trial were being selected on the basis of intensity of their diseases and ages. “We include only Covid-19 patients below 50 years. Those with heart, kidney and other serious ailments aren’t allowed to be included in the trial,” he added.
Prof Umar said that a total of 15 doctors working in different specialties and faculties at AMC and ATH were taking part in the process to win honour for the country.
https://www.dawn.com/news/1547911/abbottabad-hospital-to-conduct-drug-trials-on-covid-19-patients
Robert
Test Star
- Joined
- Nov 4, 2007
- Runs
- 37,604
- Post of the Week
- 1
What if this virus or disease is untreatable?
Treatment involves keeping the patient alive until their immune system learns the virus and they recover.
A vaccine will be found and we will eventually all get the jab and be safe.
- Joined
- Oct 2, 2004
- Runs
- 218,134
Sindh CM constitutes committee to monitor use of plasma in treating Covid-19 patients
According to a notification issued by Sindh Secretary of Health Zahid Ali Abbasi, Chief Minister Murad Ali Shah has constituted a committee to monitor the use of plasma for passive immunisation in treating Covid-19 patients.
The committee, which consists of health officials and experts, will submit a report on the efficacy and availability of plasma treatment for Covid-19 patients.
According to a notification issued by Sindh Secretary of Health Zahid Ali Abbasi, Chief Minister Murad Ali Shah has constituted a committee to monitor the use of plasma for passive immunisation in treating Covid-19 patients.
The committee, which consists of health officials and experts, will submit a report on the efficacy and availability of plasma treatment for Covid-19 patients.
Arsal_AK
ODI Debutant
- Joined
- Jan 5, 2013
- Runs
- 9,839
There is a vaccine for polio. HIV not yet but we will get there soon.
The good thing is that we know a lot about Sars-COV-2 due to early work on a SARS-COV. And the virus unlike the influenza virus doesn't mutate as quickly, so a vaccine will be quite effective against all strains we have so far.
Finally, there has never been a situation like this before. We have never had so many people working with tons of resources on a singular thing. There are 20 or so vaccines in development using novel and old techniques, with 5 in phase 1 trails already. There will be a vaccine. Just will take a year at the earliest.
In the meantime, we are getting better at understanding the mechanism of the virus. There are clinical trials going on with results expected by the end of April. Hopefully they find something effective.
By September, we will probably get antibody treatments (very promising research going on) which should be good for a 2-3 weeks of immunity (to be used for serious cases at first and then for general compassionate use). That will help turn the tide against a possible second wave as we wait for a vaccine.
The good thing is that we know a lot about Sars-COV-2 due to early work on a SARS-COV. And the virus unlike the influenza virus doesn't mutate as quickly, so a vaccine will be quite effective against all strains we have so far.
Finally, there has never been a situation like this before. We have never had so many people working with tons of resources on a singular thing. There are 20 or so vaccines in development using novel and old techniques, with 5 in phase 1 trails already. There will be a vaccine. Just will take a year at the earliest.
In the meantime, we are getting better at understanding the mechanism of the virus. There are clinical trials going on with results expected by the end of April. Hopefully they find something effective.
By September, we will probably get antibody treatments (very promising research going on) which should be good for a 2-3 weeks of immunity (to be used for serious cases at first and then for general compassionate use). That will help turn the tide against a possible second wave as we wait for a vaccine.
Abdullah719
T20I Captain
- Joined
- Apr 16, 2013
- Runs
- 44,825
Dow University experts claim they have coronavirus cure
KARACHI: Dow University of Health Sciences (DUHS) medical experts have claimed on Monday that they have created a drug to treat patients suffering from the coronavirus disease, ARY News reported.
As per details, medical experts from the DUHS claimed that Intravenous Immunoglobulin (IVIG) can cure the COVID-19 patients.
Talking to ARY News Professor Muhammad Saeed Qureshi, member of the research team, said that Intravenous Immunoglobulin is a great development in the fight against novel coronavirus.
He said the globulin was manufactured with purified antibodies, obtained from the recovered coronavirus patients. Professor Shaukat Ali, head of the research team, has termed the corona cure as a ray of hope amid the pandemic crisis.
On April 09, the Drug Regulatory Authority of Pakistan (DRAP) allowed clinical trials of blood plasma therapy to treat coronavirus patients in the country.
Plasma, the fluid in blood teeming with antibodies post-illness, has already proven effective in small studies to treat infectious diseases including Ebola and SARS.
The announcement was made by Special Assistant to PM on Health, Dr Zafar Mirza, here today. “The DRAP has also approved local manufacturing of the raw material of the anti-malarial drug, chloroquine.”
As of April 13, confirmed coronavirus cases reached 5,374 in the country as 93 people have died from the infection till to date, according to the national dashboard.
According to the available statistics, 336 cases have been reported in the country during the last 24 hours.
Punjab has the highest number of infections up to 2,594, 1,411 in Sindh, 744 in Khyber Pakhtunkhwa (KP), 230 in Balochistan, 224 in Gilgit-Baltistan (GB), 131 in Islamabad and 40 in Azad Jammu and Kashmir.
https://arynews.tv/en/dow-university-experts-claim-they-have-coronavirus-cure/
KARACHI: Dow University of Health Sciences (DUHS) medical experts have claimed on Monday that they have created a drug to treat patients suffering from the coronavirus disease, ARY News reported.
As per details, medical experts from the DUHS claimed that Intravenous Immunoglobulin (IVIG) can cure the COVID-19 patients.
Talking to ARY News Professor Muhammad Saeed Qureshi, member of the research team, said that Intravenous Immunoglobulin is a great development in the fight against novel coronavirus.
He said the globulin was manufactured with purified antibodies, obtained from the recovered coronavirus patients. Professor Shaukat Ali, head of the research team, has termed the corona cure as a ray of hope amid the pandemic crisis.
On April 09, the Drug Regulatory Authority of Pakistan (DRAP) allowed clinical trials of blood plasma therapy to treat coronavirus patients in the country.
Plasma, the fluid in blood teeming with antibodies post-illness, has already proven effective in small studies to treat infectious diseases including Ebola and SARS.
The announcement was made by Special Assistant to PM on Health, Dr Zafar Mirza, here today. “The DRAP has also approved local manufacturing of the raw material of the anti-malarial drug, chloroquine.”
As of April 13, confirmed coronavirus cases reached 5,374 in the country as 93 people have died from the infection till to date, according to the national dashboard.
According to the available statistics, 336 cases have been reported in the country during the last 24 hours.
Punjab has the highest number of infections up to 2,594, 1,411 in Sindh, 744 in Khyber Pakhtunkhwa (KP), 230 in Balochistan, 224 in Gilgit-Baltistan (GB), 131 in Islamabad and 40 in Azad Jammu and Kashmir.
https://arynews.tv/en/dow-university-experts-claim-they-have-coronavirus-cure/
- Joined
- Oct 2, 2004
- Runs
- 218,134
Heart woes spur partial stop of malaria drug study for virus
Scientists in Brazil have stopped part of a study of a malaria drug touted as a possible coronavirus treatment after heart rhythm problems developed in one-quarter of people given the higher of two doses being tested.
Chloroquine and a newer, similar drug called hydroxychloroquine, have been pushed by President Donald Trump after some very small, early tests suggested the drugs might curb the virus from entering cells. But the drugs have long been known to have potentially serious side effects, including altering the heartbeat in a way that could lead to sudden death.
Scientists in Brazil have stopped part of a study of a malaria drug touted as a possible coronavirus treatment after heart rhythm problems developed in one-quarter of people given the higher of two doses being tested.
Chloroquine and a newer, similar drug called hydroxychloroquine, have been pushed by President Donald Trump after some very small, early tests suggested the drugs might curb the virus from entering cells. But the drugs have long been known to have potentially serious side effects, including altering the heartbeat in a way that could lead to sudden death.
Heart woes spur partial stop of malaria drug study for virus
Scientists in Brazil have stopped part of a study of a malaria drug touted as a possible coronavirus treatment after heart rhythm problems developed in one-quarter of people given the higher of two doses being tested.
Chloroquine and a newer, similar drug called hydroxychloroquine, have been pushed by President Donald Trump after some very small, early tests suggested the drugs might curb the virus from entering cells. But the drugs have long been known to have potentially serious side effects, including altering the heartbeat in a way that could lead to sudden death.
They used older version of the drug Chloroquine and not the newer and safer version hydroxychloroquine HCQ. We have used HCQ on hundreds of hospitalized patients and when not used with other drugs causing heart problem, this drug is quite safe, have not seen any significant issue.
Whether its effective or not, its still a question.
JaDed
T20I Captain
- Joined
- May 5, 2014
- Runs
- 41,258
They used older version of the drug Chloroquine and not the newer and safer version hydroxychloroquine HCQ. We have used HCQ on hundreds of hospitalized patients and when not used with other drugs causing heart problem, this drug is quite safe, have not seen any significant issue.
Whether its effective or not, its still a question.
Is your hospital using Remdesvir on compassionate basis?
BoomBoomCricket
First Class Captain
- Joined
- Oct 25, 2009
- Runs
- 5,834
- Post of the Week
- 2
China has approved early-stage human tests for two experimental vaccines to combat the new coronavirus as it battles to contain imported cases, especially from neighbouring Russia, the new "front line" in the war on COVID-19.
- Joined
- Oct 2, 2004
- Runs
- 218,134
AstraZeneca Plc will start testing one of its new cancer medicines, Calquence, to see whether it can quell the excessive immune response Covid-19 triggers in some patients.
The treatment for lymphoma may lessen the severity of respiratory distress by reducing the inflammation caused by the new coronavirus, the Cambridge, England-based drugmaker said Tuesday in a statement.
Calquence will be tested on hospitalized Covid-19 patients, including some in intensive care, in a large randomized trial. The first participants will be recruited in the U.S. and several European countries in coming days, Astra said.
The U.K. drugmaker hopes “to demonstrate that adding Calquence to best supportive care reduces the need to place patients on ventilators and improves their chances of survival,” Jose Baselga, who heads cancer research at Astra, said in the statement.
The medicine targets a protein known as BTK that regulates inflammation. In some patients, Covid-19 triggers an extreme immune reaction called a cytokine storm, which can be deadly.
Astra gained as much as 7.3% in early London trading, while the FTSE 100 Index rose as much as 0.9%. Astra had said over the long U.K. holiday weekend that Koselugo, a drug for a rare, painful childhood condition, was approved in the U.S., and that Tagrisso appeared to work so well as adjuvant therapy in certain lung cancer patients a study investigating its effect was stopped early.
https://www.bloomberg.com/news/arti...-cancer-medicine-for-lethal-covid-19-reaction
The treatment for lymphoma may lessen the severity of respiratory distress by reducing the inflammation caused by the new coronavirus, the Cambridge, England-based drugmaker said Tuesday in a statement.
Calquence will be tested on hospitalized Covid-19 patients, including some in intensive care, in a large randomized trial. The first participants will be recruited in the U.S. and several European countries in coming days, Astra said.
The U.K. drugmaker hopes “to demonstrate that adding Calquence to best supportive care reduces the need to place patients on ventilators and improves their chances of survival,” Jose Baselga, who heads cancer research at Astra, said in the statement.
The medicine targets a protein known as BTK that regulates inflammation. In some patients, Covid-19 triggers an extreme immune reaction called a cytokine storm, which can be deadly.
Astra gained as much as 7.3% in early London trading, while the FTSE 100 Index rose as much as 0.9%. Astra had said over the long U.K. holiday weekend that Koselugo, a drug for a rare, painful childhood condition, was approved in the U.S., and that Tagrisso appeared to work so well as adjuvant therapy in certain lung cancer patients a study investigating its effect was stopped early.
https://www.bloomberg.com/news/arti...-cancer-medicine-for-lethal-covid-19-reaction
Is your hospital using Remdesvir on compassionate basis?
Yes we have been using it on a limited number of cases. Most of the time we got it too late . So far no dramatic effect, but its too early and too few cases we have used in to make a statement.
JaDed
T20I Captain
- Joined
- May 5, 2014
- Runs
- 41,258
Yes we have been using it on a limited number of cases. Most of the time we got it too late . So far no dramatic effect, but its too early and too few cases we have used in to make a statement.
Thanks guess it's just PR about it then for now.
- Joined
- Oct 2, 2004
- Runs
- 218,134
The number of confirmed coronavirus cases in Spain has risen by almost 5,100 in 24 hours to reach 177,633 - the country's highest single-day rise in infections in five days.
However, the daily death toll has fallen slightly, with 523 new deaths recorded.
More than 18,500 people have died with coronavirus so far in Spain.
The new figures were released as Prime Pedro Sánchez told parliament that life would not return to normal until a vaccine is found.
Earlier this week Spain began to ease some of its restrictions, with manufacturing and construction among the sectors allowed to resume work, subject to strict safety guidelines.
However, the daily death toll has fallen slightly, with 523 new deaths recorded.
More than 18,500 people have died with coronavirus so far in Spain.
The new figures were released as Prime Pedro Sánchez told parliament that life would not return to normal until a vaccine is found.
Earlier this week Spain began to ease some of its restrictions, with manufacturing and construction among the sectors allowed to resume work, subject to strict safety guidelines.
Kroll
ODI Debutant
- Joined
- Jun 4, 2017
- Runs
- 10,274
- Post of the Week
- 1
Thought it was bit of an elaborate claim, but this Dr. Shamsi might actually have been onto something.
It seems the above is similar to this:
Coronavirus: Hospital to trial 'glimmer of hope' blood treatment
A potential new treatment for coronavirus being trialled at a hospital in Wales offers patients a "glimmer of hope", specialists say.
The University Hospital of Wales (UHW) in Cardiff hopes to offer the treatment as part of a study within a month.
Blood will be extracted from people who have recovered from Covid-19 and the plasma will be given to patients.
It is hoped antibodies in the plasma of the blood could help others struggling to fight the infection.
At this early stage, the plan is to trial the treatment on patients who are severely affected by coronavirus, according to Dr Stuart Walker, medical director at Cardiff and Vale University Health Board.
"At the moment there are no other treatments per se for the viral infection itself, so this does give us a glimmer of hope," he explained.
"When you have an illness like this you produce a response in the form of antibodies in the blood stream.
"Those antibodies can potentially negate the effects of the virus in people who are suffering from it in a more severe way."
Public Health Wales will identify and write to potential donors, with the plasma collected and processed by the Welsh Blood Service.
Donors will need to have tested positive for Covid-19 and now be fully recovered.
For years, so-called "convalescent plasma" has been used on a daily basis in the health service to help combat other viruses, as well as internationally in response to the Severe Acute Respiratory Syndrome (Sars) and Ebola epidemics.
The BBC has been told other hospitals across the UK are also looking to trial the treatment, with further announcements understood to be imminent.
But UHW is "at the forefront", in part due to the expertise of its staff, according to Dr Richard Skone, clinical board director for specialist services.
"We're very fortunate here in Cardiff to have a number of specialists who have already been working in this area," he said.
"This is in the research stage at the moment but there's a chance it could help people who can't defend themselves against the virus - and for those people it could make a big difference."
The Welsh Government said Wales was playing "a leading role in the UK programme" for treating coronavirus patients using convalescent plasma.
"If the practical application works then we should be in a position where we can have a more effective response to people who are seriously ill," Health Minister Vaughan Gething said.
"And we know there are hundreds of people who are seriously ill across Wales.
"This is a really good news story for Wales and we need some good news at this really difficult time.
"People should be really proud of this work that is being done and lead from Wales."
https://www.bbc.co.uk/news/uk-wales-52299404
Abdullah719
T20I Captain
- Joined
- Apr 16, 2013
- Runs
- 44,825
Report: Covid-19 patients recovering quickly after getting experimental drug remdesivir
https://edition.cnn.com/2020/04/16/health/coronavirus-remdesivir-trial/index.html
(CNN) Covid-19 patients who are getting an experimental drug called remdesivir have been recovering quickly, with most going home in days, STAT News reported Thursday after it obtained a video of a conversation about the trial.
The patients taking part in a clinical trial of the drug have all had severe respiratory symptoms and fever, but were able to leave the hospital after less than a week of treatment, STAT quoted the doctor leading the trial as saying.
"The best news is that most of our patients have already been discharged, which is great. We've only had two patients perish," Dr. Kathleen Mullane, an infectious disease specialist at the University of Chicago who is leading the clinical trial, said in the video.
Mullane did not immediately respond to a request for comment from CNN.
The University of Chicago said Mullane's comments constituted partial information.
"Partial data from an ongoing clinical trial is by definition incomplete and should never be used to draw conclusions about the safety or efficacy of a potential treatment that is under investigation," it said in a statement.
"In this case, information from an internal forum for research colleagues concerning work in progress was released without authorization. Drawing any conclusions at this point is premature and scientifically unsound."
There is no approved therapy for the Covid-19, which can cause severe pneumonia and acute respiratory distress syndrome in some patients. But the National Institutes of Health is organizing trials of several drugs and other treatments, among them remdesivir.
The drug, made by Gilead Sciences, was tested against Ebola with little success, but multiple studies in animals showed the drug could both prevent and treat coronaviruses related to Covid-19, including SARS (Severe Acute Respiratory Syndrome) and MERS (Middle East Respiratory Syndrome).
Back in February, the World Health Organization said remdesivir showed potential against Covid-19.
STAT said it obtained and viewed a copy of the video discussion Mullane had last week with colleagues about the trial.
"Most of our patients are severe and most of them are leaving at six days, so that tells us duration of therapy doesn't have to be 10 days," she was quoted as saying.
However, the trial does not include what's known as a control group, so it will be difficult to say whether the drug is truly helping patients recover better. With a control arm, some patients do not receive the drug being tested so that doctors can determine whether it's the drug that is really affecting their condition.
Trials of the drug are ongoing at dozens of other clinical centers, as well. Gilead is sponsoring tests of the drug in 2,400 patients with severe Covid-19 symptoms in 152 trial sites around the world. It's also testing the drug in 1,600 patients with moderate symptoms at 169 hospitals and clinics around the world.
Gilead said it expected results from the trial by the end of the month.
"We understand the urgent need for a COVID-19 treatment and the resulting interest in data on our investigational antiviral drug remdesivir," the company said in a statement to CNN. But it said a few stories about patients are just that -- stories.
"The totality of the data need to be analyzed in order to draw any conclusions from the trial. Anecdotal reports, while encouraging, do not provide the statistical power necessary to determine the safety and efficacy profile of remdesivir as a treatment for Covid-19," Gilead said.
The patients taking part in a clinical trial of the drug have all had severe respiratory symptoms and fever, but were able to leave the hospital after less than a week of treatment, STAT quoted the doctor leading the trial as saying.
"The best news is that most of our patients have already been discharged, which is great. We've only had two patients perish," Dr. Kathleen Mullane, an infectious disease specialist at the University of Chicago who is leading the clinical trial, said in the video.
Mullane did not immediately respond to a request for comment from CNN.
The University of Chicago said Mullane's comments constituted partial information.
"Partial data from an ongoing clinical trial is by definition incomplete and should never be used to draw conclusions about the safety or efficacy of a potential treatment that is under investigation," it said in a statement.
"In this case, information from an internal forum for research colleagues concerning work in progress was released without authorization. Drawing any conclusions at this point is premature and scientifically unsound."
There is no approved therapy for the Covid-19, which can cause severe pneumonia and acute respiratory distress syndrome in some patients. But the National Institutes of Health is organizing trials of several drugs and other treatments, among them remdesivir.
The drug, made by Gilead Sciences, was tested against Ebola with little success, but multiple studies in animals showed the drug could both prevent and treat coronaviruses related to Covid-19, including SARS (Severe Acute Respiratory Syndrome) and MERS (Middle East Respiratory Syndrome).
Back in February, the World Health Organization said remdesivir showed potential against Covid-19.
STAT said it obtained and viewed a copy of the video discussion Mullane had last week with colleagues about the trial.
"Most of our patients are severe and most of them are leaving at six days, so that tells us duration of therapy doesn't have to be 10 days," she was quoted as saying.
However, the trial does not include what's known as a control group, so it will be difficult to say whether the drug is truly helping patients recover better. With a control arm, some patients do not receive the drug being tested so that doctors can determine whether it's the drug that is really affecting their condition.
Trials of the drug are ongoing at dozens of other clinical centers, as well. Gilead is sponsoring tests of the drug in 2,400 patients with severe Covid-19 symptoms in 152 trial sites around the world. It's also testing the drug in 1,600 patients with moderate symptoms at 169 hospitals and clinics around the world.
Gilead said it expected results from the trial by the end of the month.
"We understand the urgent need for a COVID-19 treatment and the resulting interest in data on our investigational antiviral drug remdesivir," the company said in a statement to CNN. But it said a few stories about patients are just that -- stories.
"The totality of the data need to be analyzed in order to draw any conclusions from the trial. Anecdotal reports, while encouraging, do not provide the statistical power necessary to determine the safety and efficacy profile of remdesivir as a treatment for Covid-19," Gilead said.
Arsal_AK
ODI Debutant
- Joined
- Jan 5, 2013
- Runs
- 9,839
Yeah remdesivir is super promising. The bad news is unless they lisence it out, there won't be enough of it.
Another hopeful is Ivermectin, astonishing first results so let's see with a more controlled trail. Amazing in vitro results too.
Another hopeful is Ivermectin, astonishing first results so let's see with a more controlled trail. Amazing in vitro results too.
- Joined
- Oct 2, 2004
- Runs
- 218,134
Rapid testing kit' can confirm if you have Covid-19 in 10 minutes
Sindh Chief Minister's Special Assistant Waqar Mehdi has expressed concern over the increasing number of cases emerging in Karachi's South district. During a meeting of the monitoring committee for district South, it was decided that rapid testing be conducted in areas where most were being reported from.
The participants of the meeting were told that a rapid testing kit has been procured by the provincial government, which would confirm the presence of the virus in 10 minutes. The kit was used to conduct Mehdi's test, which turned out to be negative.
Sindh Chief Minister's Special Assistant Waqar Mehdi has expressed concern over the increasing number of cases emerging in Karachi's South district. During a meeting of the monitoring committee for district South, it was decided that rapid testing be conducted in areas where most were being reported from.
The participants of the meeting were told that a rapid testing kit has been procured by the provincial government, which would confirm the presence of the virus in 10 minutes. The kit was used to conduct Mehdi's test, which turned out to be negative.
- Joined
- Oct 2, 2004
- Runs
- 218,134
When will we have a Covid-19 vaccine? Public-facing scientists such as the UK’s chief scientific adviser, Sir Patrick Vallance, and his US counterpart, Anthony Fauci, keep repeating that it won’t be before 12 to 18 months. But other voices – including some of those in the race to create a vaccine themselves – have suggested that it could be as early as June. Who is right?
The former, probably, but it’s complicated because this pandemic is forcing change at almost every step in the process by which a new vaccine arrives at a needle near us.
“It really depends on what you mean by ‘having a vaccine’,” says Marian Wentworth, president and CEO of Management Sciences for Health, a Massachusetts-based global not-for-profit organisation that seeks to build resilient health systems, and a long-time observer of vaccine development. “If you mean one that can be used in a mass vaccination campaign, allowing us all to get on with our lives, then 12 to 18 months is probably right.”
But in terms of an experimental vaccine that is deemed safe and effective enough to be rolled out in a more limited way – to high-risk groups such as health workers, say – that could be ready within weeks or months, under emergency rules developed by drug regulatory agencies and the World Health Organization in the context of the recent Ebola epidemics in Africa.
When the University of Oxford’s Adrian Hill told the Guardian that his group’s Covid-19 vaccine candidate could be ready by the summer, it was this kind of readiness to which he was probably referring. The group, led by Sarah Gilbert, has since stated that a vaccine shown to be effective in phase-3 clinical trials that could be manufactured in large quantities won’t be ready before the autumn even in a best-case scenario. And that scenario is “highly ambitious and subject to change”.
Normally, a vaccine is developed in the lab before being tested on animals. If it proves safe and generates a promising immune response in this pre-clinical phase, it enters human or clinical trials. These are divided into three phases, each of which takes longer and involves more people than the previous one. Phase 1 establishes the vaccine’s safety in a small group of healthy individuals, with the goal of ruling out debilitating side effects. Phases 2 and 3 test efficacy, and in an outbreak like the present one they are conducted in places where the disease is prevalent. In parallel with these later phases, production capacity for the candidate vaccine is gradually built up, so that factories are capable of producing it on a large scale if and when regulatory agencies judge that it should be licensed.
In an article published in The New England Journal of Medicine on 30 March, representatives of the Oslo-based not-for-profit Coalition for Epidemic Preparedness Innovations (Cepi), which is helping to finance and coordinate Covid-19 vaccine development, laid out an accelerated version of this process that they believe is more suited to a pandemic. This “pandemic paradigm” implements certain steps in parallel, such as animal and phase-1 clinical testing. It also involves scaling up production capacity before sufficient safety and efficacy data are available – a financially risky step, given that that may never materialise, and one that requires governments and not-for-profit organisations such as Cepi to share that extra financial risk with pharmaceutical companies if they want them to engage. Mass production is critical in a pandemic, when hundreds of millions if not billions of doses are needed – and many countries are now scrambling to build new vaccine production facilities.
“People now appreciate that the lengthy process of conventional licensing of vaccines is not going to be helpful in the context of an epidemic,” says Beate Kampmann, who heads the vaccine centre at the London School of Hygiene and Tropical Medicine.
Prudently, Cepi did not attach a timeline to its accelerated paradigm, but the 12- to 18-month estimate already takes it into account. Bringing a new vaccine to the clinic has taken 10 to 20 years in the past. Nevertheless, the accelerated paradigm is being implemented now. A Boston-based biotech firm, Moderna, saw its experimental Covid-19 vaccine enter human trials on 16 March, just 10 weeks after the first genetic sequences of Sars-CoV-2 – the virus that causes the disease – were released. Others will follow soon.
“We’re getting to candidates much more quickly,” says Kampmann, who puts this progress down to advances made in the fight against Ebola. “The step-up in technology that we have seen in the last five years has really made a difference.”
There are many hurdles ahead, though. Most of the 70-odd Covid-19 vaccine candidates being developed and tested will not make it to the licensing stage, and those that have been fastest out of the blocks may still encounter problems later on. Moderna’s innovative technology allowed it to generate a candidate quickly, but no vaccine using this platform has been licensed to date.
At the Pasteur Institute in Paris, on the other hand, a Covid-19 vaccine candidate is still in pre-clinical development, but because it piggybacks on established technology – a licensed measles vaccine – the testing and licensing processes will go faster. And this kind of vaccine can already be produced in large quantities.
While there can be no shortcuts to establishing safety and efficacy, proposals have been put forward for how these experimental vaccines might be tested more rapidly without sacrificing scientific rigour. In February, for example, the WHO published a draft protocol for phase 2 and 3 trials that would test a number of candidates simultaneously, in multi-country trials according to standardised criteria.
Another proposal is to conduct controlled human challenge trials, in which healthy volunteers are given a candidate vaccine and then infected with Sars-CoV-2. These are ethically questionable, especially before scientists understand why young and otherwise healthy people are ending up on ventilators. A similar approach, being implemented by the London-based clinical research group Hvivo, invites volunteers to be infected with a milder coronavirus – but how applicable its findings will be to Sars-CoV-2 is not clear.
There are still many unknowns with respect to Covid-19, including for how long any vaccine will provide protection. A strong indication of this will be whether people who have recovered from the disease can catch it again. There have been anecdotal reports of re-infection, but the phenomenon is not well understood. “If our own body can’t prevent us from getting it again, that would be one pretty damning signal,” says Wentworth.
Once a vaccine is licensed, there will still be political obstacles to getting it to where it’s needed, because each country or public health jurisdiction has to make its own decision to roll it out. There will also be issues of prioritisation – who should get it first, if supplies are limited – which authorities are discussing now.
A vaccine that is approved a year from now may arrive after the end of the current pandemic, but if so it won’t be wasted – first because Covid-19 may recur seasonally, and second because the vaccine could itself be repurposed in the event of an outbreak of a different coronavirus. That will be no consolation to victims of this pandemic, or their relatives, but it does mean that humanity will be better protected in future. As Wentworth says: “That learning, we won’t unlearn.”
https://www.theguardian.com/world/2020/apr/19/coronavirus-vaccine-when-will-we-have-one
The former, probably, but it’s complicated because this pandemic is forcing change at almost every step in the process by which a new vaccine arrives at a needle near us.
“It really depends on what you mean by ‘having a vaccine’,” says Marian Wentworth, president and CEO of Management Sciences for Health, a Massachusetts-based global not-for-profit organisation that seeks to build resilient health systems, and a long-time observer of vaccine development. “If you mean one that can be used in a mass vaccination campaign, allowing us all to get on with our lives, then 12 to 18 months is probably right.”
But in terms of an experimental vaccine that is deemed safe and effective enough to be rolled out in a more limited way – to high-risk groups such as health workers, say – that could be ready within weeks or months, under emergency rules developed by drug regulatory agencies and the World Health Organization in the context of the recent Ebola epidemics in Africa.
When the University of Oxford’s Adrian Hill told the Guardian that his group’s Covid-19 vaccine candidate could be ready by the summer, it was this kind of readiness to which he was probably referring. The group, led by Sarah Gilbert, has since stated that a vaccine shown to be effective in phase-3 clinical trials that could be manufactured in large quantities won’t be ready before the autumn even in a best-case scenario. And that scenario is “highly ambitious and subject to change”.
Normally, a vaccine is developed in the lab before being tested on animals. If it proves safe and generates a promising immune response in this pre-clinical phase, it enters human or clinical trials. These are divided into three phases, each of which takes longer and involves more people than the previous one. Phase 1 establishes the vaccine’s safety in a small group of healthy individuals, with the goal of ruling out debilitating side effects. Phases 2 and 3 test efficacy, and in an outbreak like the present one they are conducted in places where the disease is prevalent. In parallel with these later phases, production capacity for the candidate vaccine is gradually built up, so that factories are capable of producing it on a large scale if and when regulatory agencies judge that it should be licensed.
In an article published in The New England Journal of Medicine on 30 March, representatives of the Oslo-based not-for-profit Coalition for Epidemic Preparedness Innovations (Cepi), which is helping to finance and coordinate Covid-19 vaccine development, laid out an accelerated version of this process that they believe is more suited to a pandemic. This “pandemic paradigm” implements certain steps in parallel, such as animal and phase-1 clinical testing. It also involves scaling up production capacity before sufficient safety and efficacy data are available – a financially risky step, given that that may never materialise, and one that requires governments and not-for-profit organisations such as Cepi to share that extra financial risk with pharmaceutical companies if they want them to engage. Mass production is critical in a pandemic, when hundreds of millions if not billions of doses are needed – and many countries are now scrambling to build new vaccine production facilities.
“People now appreciate that the lengthy process of conventional licensing of vaccines is not going to be helpful in the context of an epidemic,” says Beate Kampmann, who heads the vaccine centre at the London School of Hygiene and Tropical Medicine.
Prudently, Cepi did not attach a timeline to its accelerated paradigm, but the 12- to 18-month estimate already takes it into account. Bringing a new vaccine to the clinic has taken 10 to 20 years in the past. Nevertheless, the accelerated paradigm is being implemented now. A Boston-based biotech firm, Moderna, saw its experimental Covid-19 vaccine enter human trials on 16 March, just 10 weeks after the first genetic sequences of Sars-CoV-2 – the virus that causes the disease – were released. Others will follow soon.
“We’re getting to candidates much more quickly,” says Kampmann, who puts this progress down to advances made in the fight against Ebola. “The step-up in technology that we have seen in the last five years has really made a difference.”
There are many hurdles ahead, though. Most of the 70-odd Covid-19 vaccine candidates being developed and tested will not make it to the licensing stage, and those that have been fastest out of the blocks may still encounter problems later on. Moderna’s innovative technology allowed it to generate a candidate quickly, but no vaccine using this platform has been licensed to date.
At the Pasteur Institute in Paris, on the other hand, a Covid-19 vaccine candidate is still in pre-clinical development, but because it piggybacks on established technology – a licensed measles vaccine – the testing and licensing processes will go faster. And this kind of vaccine can already be produced in large quantities.
While there can be no shortcuts to establishing safety and efficacy, proposals have been put forward for how these experimental vaccines might be tested more rapidly without sacrificing scientific rigour. In February, for example, the WHO published a draft protocol for phase 2 and 3 trials that would test a number of candidates simultaneously, in multi-country trials according to standardised criteria.
Another proposal is to conduct controlled human challenge trials, in which healthy volunteers are given a candidate vaccine and then infected with Sars-CoV-2. These are ethically questionable, especially before scientists understand why young and otherwise healthy people are ending up on ventilators. A similar approach, being implemented by the London-based clinical research group Hvivo, invites volunteers to be infected with a milder coronavirus – but how applicable its findings will be to Sars-CoV-2 is not clear.
There are still many unknowns with respect to Covid-19, including for how long any vaccine will provide protection. A strong indication of this will be whether people who have recovered from the disease can catch it again. There have been anecdotal reports of re-infection, but the phenomenon is not well understood. “If our own body can’t prevent us from getting it again, that would be one pretty damning signal,” says Wentworth.
Once a vaccine is licensed, there will still be political obstacles to getting it to where it’s needed, because each country or public health jurisdiction has to make its own decision to roll it out. There will also be issues of prioritisation – who should get it first, if supplies are limited – which authorities are discussing now.
A vaccine that is approved a year from now may arrive after the end of the current pandemic, but if so it won’t be wasted – first because Covid-19 may recur seasonally, and second because the vaccine could itself be repurposed in the event of an outbreak of a different coronavirus. That will be no consolation to victims of this pandemic, or their relatives, but it does mean that humanity will be better protected in future. As Wentworth says: “That learning, we won’t unlearn.”
https://www.theguardian.com/world/2020/apr/19/coronavirus-vaccine-when-will-we-have-one
Kroll
ODI Debutant
- Joined
- Jun 4, 2017
- Runs
- 10,274
- Post of the Week
- 1
Coronavirus: Plasma treatment to be trialled
The UK is gearing up to use the blood of coronavirus survivors to treat hospital patients ill with the disease.
NHS Blood and Transplant is asking some people who recovered from Covid-19 to donate blood so they can potentially assess the therapy in trials.
The hope is that the antibodies they have built up will help to clear the virus in others.
The US has already started a major project to study this, involving more than 1,500 hospitals.
When a person has Covid-19, their immune system responds by creating antibodies, which attack the virus.
Over time these build up and can be found in the plasma, the liquid portion of the blood.
NHSBT is now approaching patients who have recovered from Covid-19 to see if plasma from them can be given to people who are currently ill with the virus.
A statement from the organisation said: "We envisage that this will be initially used in trials as a possible treatment for Covid-19.
"If fully approved, the trials will investigate whether convalescent plasma transfusions could improve a Covid-19 patient's speed of recovery and chances of survival.
"All clinical trials have to follow a rigorous approval process to protect patients and to ensure robust results are generated. We are working closely with the government and all relevant bodies to move through the approvals process as quickly as possible."
Are other groups doing this?
Several groups in the UK have been looking into using blood plasma.
University Hospital of Wales (UHW) in Cardiff announced this week that it wanted to trial the technology.
Professor Sir Robert Lechler, president of the Academy of Medical Sciences and executive director of King's Health Partners, which includes King's College London and three major London hospitals, is also hoping to set up another small-scale trial.
He wants to use plasma for seriously ill patients that have no other treatment options, while a larger national trial is getting under way.
He said: "I would be disappointed if we weren't able to see some patients given this form of therapy within a couple of weeks. Let's hope that the NHSBT national trial gets into gear really quickly."
He said the UK had moved slowly to test the treatment.
"I think there are many aspects of this pandemic we'll look back on and say, I wonder why we didn't move a little bit faster. I think this could be one of those".
What is the situation in the rest of the world?
Around the world, trials are now under way to look at using plasma.
In just three weeks, scientists in the US have organised a nationwide project and about 600 patients have been treated so far.
Prof Michael Joyner, from the Mayo Clinic, is leading the work.
He said: "The thing we've learned in the first week of administration is that no major safety signals have emerged and administration of the product does not appear to be causing a whole lot of unanticipated side effects.
"There are anecdotal reports of oxygenation improving and other patient improvements. Those are certainly heartening, but they need to be rigorously evaluated."
He said the therapy was "rough and ready".
"There's a lot we don't understand about the plasma. We're going to learn more about what's in the plasma, the components, the antibody levels, and other factors that may be there as the weeks go on.
"But sometimes, as a physician, you just have to try to take a shot on goal when you have a shot."
How has this approach helped in previous pandemics?
Harnessing the blood of recovered patients is not a new idea in medicine. It was used more than 100 years ago during the Spanish Flu epidemic, and more recently for Ebola and Sars.
So far, only small studies have looked at its efficacy, and there is a great deal of research that needs to be done to see how effective it will be against coronavirus.
But in the US, they say they have no shortage of people who want to help.
Bruce Sachais, chief medical officer at the New York Blood Center, said: "People have been amazing. They keep coming out in droves.
"We have hundreds and hundreds of donors and we've been able to collect over 1,000 units already. It's really heart-warming to see that for people who have gone through the infection, in various degrees, their main concern is 'how can I now help others?'"
Scientists say plasma won't be a magic bullet.
But while our options for treating coronavirus are so limited, the hope is it could help until a vaccine is found.
https://www.bbc.co.uk/news/health-52348368
The UK is gearing up to use the blood of coronavirus survivors to treat hospital patients ill with the disease.
NHS Blood and Transplant is asking some people who recovered from Covid-19 to donate blood so they can potentially assess the therapy in trials.
The hope is that the antibodies they have built up will help to clear the virus in others.
The US has already started a major project to study this, involving more than 1,500 hospitals.
When a person has Covid-19, their immune system responds by creating antibodies, which attack the virus.
Over time these build up and can be found in the plasma, the liquid portion of the blood.
NHSBT is now approaching patients who have recovered from Covid-19 to see if plasma from them can be given to people who are currently ill with the virus.
A statement from the organisation said: "We envisage that this will be initially used in trials as a possible treatment for Covid-19.
"If fully approved, the trials will investigate whether convalescent plasma transfusions could improve a Covid-19 patient's speed of recovery and chances of survival.
"All clinical trials have to follow a rigorous approval process to protect patients and to ensure robust results are generated. We are working closely with the government and all relevant bodies to move through the approvals process as quickly as possible."
Are other groups doing this?
Several groups in the UK have been looking into using blood plasma.
University Hospital of Wales (UHW) in Cardiff announced this week that it wanted to trial the technology.
Professor Sir Robert Lechler, president of the Academy of Medical Sciences and executive director of King's Health Partners, which includes King's College London and three major London hospitals, is also hoping to set up another small-scale trial.
He wants to use plasma for seriously ill patients that have no other treatment options, while a larger national trial is getting under way.
He said: "I would be disappointed if we weren't able to see some patients given this form of therapy within a couple of weeks. Let's hope that the NHSBT national trial gets into gear really quickly."
He said the UK had moved slowly to test the treatment.
"I think there are many aspects of this pandemic we'll look back on and say, I wonder why we didn't move a little bit faster. I think this could be one of those".
What is the situation in the rest of the world?
Around the world, trials are now under way to look at using plasma.
In just three weeks, scientists in the US have organised a nationwide project and about 600 patients have been treated so far.
Prof Michael Joyner, from the Mayo Clinic, is leading the work.
He said: "The thing we've learned in the first week of administration is that no major safety signals have emerged and administration of the product does not appear to be causing a whole lot of unanticipated side effects.
"There are anecdotal reports of oxygenation improving and other patient improvements. Those are certainly heartening, but they need to be rigorously evaluated."
He said the therapy was "rough and ready".
"There's a lot we don't understand about the plasma. We're going to learn more about what's in the plasma, the components, the antibody levels, and other factors that may be there as the weeks go on.
"But sometimes, as a physician, you just have to try to take a shot on goal when you have a shot."
How has this approach helped in previous pandemics?
Harnessing the blood of recovered patients is not a new idea in medicine. It was used more than 100 years ago during the Spanish Flu epidemic, and more recently for Ebola and Sars.
So far, only small studies have looked at its efficacy, and there is a great deal of research that needs to be done to see how effective it will be against coronavirus.
But in the US, they say they have no shortage of people who want to help.
Bruce Sachais, chief medical officer at the New York Blood Center, said: "People have been amazing. They keep coming out in droves.
"We have hundreds and hundreds of donors and we've been able to collect over 1,000 units already. It's really heart-warming to see that for people who have gone through the infection, in various degrees, their main concern is 'how can I now help others?'"
Scientists say plasma won't be a magic bullet.
But while our options for treating coronavirus are so limited, the hope is it could help until a vaccine is found.
https://www.bbc.co.uk/news/health-52348368
- Joined
- Oct 2, 2004
- Runs
- 218,134
Oxford University researchers will begin human trials of a potential vaccine on Thursday, says Health Secretary Matt Hancock
==
Appeal for volunteers for UK vaccine trial
The UK has announced it is giving more than £40m for two British projects searching for a vaccine to coronavirus - with trials to start next week.
One of those projects, led by the Imperial College London's Department of Infectious Diseases, is appealing for volunteers.
In a tweet, the college's trust said it was looking for healthy people aged between 18 and 55, and that successful applicants would be paid up to £625 for taking part.
==
Appeal for volunteers for UK vaccine trial
The UK has announced it is giving more than £40m for two British projects searching for a vaccine to coronavirus - with trials to start next week.
One of those projects, led by the Imperial College London's Department of Infectious Diseases, is appealing for volunteers.
In a tweet, the college's trust said it was looking for healthy people aged between 18 and 55, and that successful applicants would be paid up to £625 for taking part.
- Joined
- Oct 2, 2004
- Runs
- 218,134
German vaccine set for human testing
A possible vaccine for Covid-19, developed by German firm BioNTech and US drug giant Pfizer, has been given the green light for human testing.
The trial will begin with 200 healthy people, aged between 18 and 55, being given the vaccine.
"This is a good sign that the development of a vaccine in Germany is so progressed that we can start with the first studies," the country's health minister, Jens Spahn, said Wednesday. "At the same time the advice remains important that it will take months. This is an injection in the body. Safety first is the guideline for such a vaccine."
Only a handful of vaccine candidates have been approved for clinical testing on humans globally, and experts caution that it will take at least a year for a viable vaccine to be developed.
Scientists at the UK's Oxford University are starting human trials of a possible vaccine this week. Other clinical trials are taking place in the US and China.
A possible vaccine for Covid-19, developed by German firm BioNTech and US drug giant Pfizer, has been given the green light for human testing.
The trial will begin with 200 healthy people, aged between 18 and 55, being given the vaccine.
"This is a good sign that the development of a vaccine in Germany is so progressed that we can start with the first studies," the country's health minister, Jens Spahn, said Wednesday. "At the same time the advice remains important that it will take months. This is an injection in the body. Safety first is the guideline for such a vaccine."
Only a handful of vaccine candidates have been approved for clinical testing on humans globally, and experts caution that it will take at least a year for a viable vaccine to be developed.
Scientists at the UK's Oxford University are starting human trials of a possible vaccine this week. Other clinical trials are taking place in the US and China.
- Joined
- Oct 2, 2004
- Runs
- 218,134
A drug championed by the US president as a possible cure for COVID-19 has been shown to provide no benefit and possibly a higher risk of death.
Hydroxychloroquine had been championed by Mr Trump after anecdotal reports that the drug, which is also used to treat lupus and rheumatoid arthritis, could help coronavirus patients.
However the new study found it might actually be harmful to those with the disease.
The American research analysed the records of 368 male patients with confirmed coronavirus.
They were all at veterans' hospitals in the US and died or were discharged by 11 April, according to a version of the study posted online.
Some 28% of 97 patients given hydroxychloroquine and standard care died, while 22% of the 113 patients who were given hydroxychloroquine and antibiotic azithromycin died.
But the death rate among the 158 patients who received only standard care - without hydroxychloroquine - was 11%.
The study, which took patients' individual characteristics into account, has not yet been accepted for publication in a medical journal but it has been submitted for expert review.
Hydroxychloroquine also appeared to have no effect on whether a patient needed breathing support: 13% of those who got the drug needed ventilation, versus 14% of those who received only supportive care.
Only 7% of those given the hydroxychloroquine and azithromycin needed help to breathe.
Dr Jeremy Falk, a pulmonary specialist at Cedars-Sinai Medical Center in Los Angeles, said the study was one of a few in the past few weeks to have placed doubt on whether hydroxychloroquine is beneficial.
Dr Falk, who was not involved in the study, said: "We were using it on just about everybody early on. Now we are using it more sparingly."
There are no proven treatments for COVID-19, but hydroxychloroquine, an old malaria drug, has been getting a lot of attention after Mr Trump promoted it as a "game changer".
"What do you have to lose? Take it," he said earlier this month, adding that - while he has no medical or scientific training - his "common sense" qualified him to make such claims.
However, because of a lack of conclusive scientific studies, the US government's top infectious disease specialist, Dr Anthony Fauci, had urged caution.
https://news.sky.com/story/coronavi...t-research-finds-11976830?dcmp=snt-sf-twitter
Hydroxychloroquine had been championed by Mr Trump after anecdotal reports that the drug, which is also used to treat lupus and rheumatoid arthritis, could help coronavirus patients.
However the new study found it might actually be harmful to those with the disease.
The American research analysed the records of 368 male patients with confirmed coronavirus.
They were all at veterans' hospitals in the US and died or were discharged by 11 April, according to a version of the study posted online.
Some 28% of 97 patients given hydroxychloroquine and standard care died, while 22% of the 113 patients who were given hydroxychloroquine and antibiotic azithromycin died.
But the death rate among the 158 patients who received only standard care - without hydroxychloroquine - was 11%.
The study, which took patients' individual characteristics into account, has not yet been accepted for publication in a medical journal but it has been submitted for expert review.
Hydroxychloroquine also appeared to have no effect on whether a patient needed breathing support: 13% of those who got the drug needed ventilation, versus 14% of those who received only supportive care.
Only 7% of those given the hydroxychloroquine and azithromycin needed help to breathe.
Dr Jeremy Falk, a pulmonary specialist at Cedars-Sinai Medical Center in Los Angeles, said the study was one of a few in the past few weeks to have placed doubt on whether hydroxychloroquine is beneficial.
Dr Falk, who was not involved in the study, said: "We were using it on just about everybody early on. Now we are using it more sparingly."
There are no proven treatments for COVID-19, but hydroxychloroquine, an old malaria drug, has been getting a lot of attention after Mr Trump promoted it as a "game changer".
"What do you have to lose? Take it," he said earlier this month, adding that - while he has no medical or scientific training - his "common sense" qualified him to make such claims.
However, because of a lack of conclusive scientific studies, the US government's top infectious disease specialist, Dr Anthony Fauci, had urged caution.
https://news.sky.com/story/coronavi...t-research-finds-11976830?dcmp=snt-sf-twitter